Glucose Detection and Measurement
Glucose aka sugar is an important source of energy in living organism. Regulating the Glucose level in the body, which is essential contributor of one’s health, is of prime interest in the medical application. There are three known technique to measure the Glucose level in one’s body, via Blood, Eye, and/or Sweat tests.
Blood test is known to be the best way to measure Glucose level as you may have seen on your annual physical test. It is an invasive technique to draw blood and assess the Glucose level. There are many handheld devices that allow the test by end users on periodic basis.
Eye test is fairly non-invasive technique that optically assesses the Glucose level. You may have experienced that when you visit your Optometrist on annual basis.
Alternative way to measure Glucose is by testing human body sweat. This is non-invasive technique which is preferred by end users. Many companies are working toward design and development of wireless technique to measure Glucose via human body sweat.
Market traction and opportunities are tremendous for Glucose Sensor and Measurement for mobile UE devices.
Partner with ORTENGA for product definitions and component requirements for Glucose detection and measurement device.
The Architecture of Non-Invasive Glucose Monitoring
Defining Accuracy, Power, and Use-Case Before Building the Biosensor
Glucose (sugar) is a primary source of energy in living organisms. Maintaining stable glucose levels is essential for metabolic health and overall physiological balance. As diabetes and metabolic disorders continue to increase globally, the demand for reliable and convenient glucose monitoring technologies continues to grow.
Monitoring glucose levels allows individuals and clinicians to understand how the body processes energy and to manage metabolic conditions effectively. Over the years, several techniques have been developed to measure glucose levels in the human body.
Today, three primary approaches are commonly explored:
- Blood-based measurement
- Eye-based optical measurement
- Sweat-based sensing
Each approach represents a different balance between accuracy, invasiveness, and system complexity.
Blood-Based Glucose Measurement
Blood testing remains the most widely accepted and clinically accurate method for glucose measurement. Most people are familiar with this method from routine medical examinations or personal glucose monitoring devices used by individuals managing diabetes.
The technique typically requires drawing a small amount of blood using a finger-prick device. The blood sample is then analyzed using a glucose meter or test strip system that measures the concentration of glucose.
Because blood directly carries glucose throughout the body, this method provides the most accurate representation of real-time glucose levels.
However, the method has several limitations. It is invasive, requires consumable test strips, and cannot easily provide continuous measurements without specialized wearable sensors.
Despite these limitations, blood-based sensing remains the clinical reference standard against which other technologies are compared.
Eye-Based Optical Glucose Detection
Another emerging approach to glucose monitoring involves optical assessment through the eye. Because the eye contains fluids that can reflect biochemical changes in the body, researchers have explored methods to estimate glucose concentration through optical measurement techniques.
These systems often rely on infrared spectroscopy, retinal imaging, or tear-fluid analysis to infer glucose levels.
Eye-based measurement has the advantage of being minimally invasive, as it does not require drawing blood. However, the relationship between glucose concentration in tear fluid or ocular tissue and blood glucose levels can vary, which introduces calibration challenges.
As a result, while promising, eye-based glucose monitoring remains an active area of research and development.
Sweat-Based Glucose Detection
An alternative method for glucose measurement involves detecting glucose concentrations in human sweat. Because sweat is naturally produced by the body, it offers an attractive pathway for non-invasive sensing.
Recent advances in wearable electronics, microfluidics, and biosensor technology have enabled the development of skin-mounted patches capable of analyzing sweat composition.
These devices typically combine:
- biochemical sensors
- microfluidic sampling channels
- signal-processing electronics
- wireless communication
The ability to integrate these components into wearable platforms opens the door to continuous and non-invasive glucose monitoring.
This approach is particularly attractive for integration with mobile user equipment (UE) devices, enabling real-time monitoring and data transmission to smartphones or cloud-based health platforms.
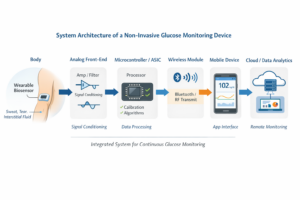
Figure 1 — System Architecture of a Non-Invasive Glucose Monitoring Device
A scalable glucose monitoring platform requires coordinated engineering across biosensing chemistry, analog electronics, embedded processing, wireless connectivity, and data analytics.
Trade Space of Glucose Measurement Techniques
Each glucose measurement method represents a different balance between accuracy, convenience, and invasiveness.
Blood testing provides the highest accuracy but requires invasive sampling. Optical methods offer reduced invasiveness but introduce calibration and reliability challenges. Sweat-based sensing offers the greatest convenience and potential for continuous monitoring, but it also introduces significant engineering complexity.
Understanding these trade-offs is essential when designing next-generation glucose monitoring systems.
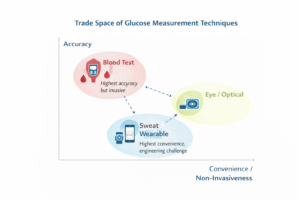
Figure 2 — Trade Space of Glucose Measurement Techniques
Blood testing provides clinical accuracy but requires invasive sampling. Emerging wearable techniques aim to deliver continuous monitoring while maintaining measurement fidelity.
System Engineering Perspective
Developing a reliable non-invasive glucose monitoring device is not simply a matter of designing a biochemical sensor. It requires coordinated engineering across multiple disciplines.
Key design considerations include:
- sensor sensitivity and noise performance
- power consumption and battery life
- wireless communication reliability
- calibration stability over time
- environmental and physiological variability
- device form factor and user comfort
In many cases, startups focus heavily on the biosensor chemistry while underestimating the complexity of the surrounding system architecture required to produce a scalable product.
Successful products require a balanced integration of sensing technology, electronics, embedded software, and wireless connectivity.
Market Opportunity
The market opportunity for glucose monitoring technologies continues to expand as healthcare shifts toward preventative monitoring and digital health platforms.
Wearable health devices, mobile integration, and cloud-based analytics are creating new pathways for continuous health monitoring beyond traditional clinical environments.
Non-invasive glucose sensing represents one of the most significant opportunities in the medical device industry, particularly for integration with wearable and mobile platforms.
However, achieving reliable and scalable solutions requires disciplined engineering and clear product definition.
ORTENGA Approach
ORTENGA supports emerging technology companies by helping define the system architecture required for scalable sensing and medical platforms through a disciplined Audit → Design → Validate methodology.
Audit
The Audit phase establishes the purpose of the system.
This phase defines:
- target use cases
- market or clinical justification
- operating environments
- user interaction models
- economic viability
- regulatory considerations
At this stage, the goal is not to define engineering specifications, but to clearly articulate why the system should exist and what problems it must solve.
Without clearly defined use cases and business justification, engineering teams risk optimizing technology without delivering meaningful products.
Design
The Design phase translates the use cases identified during the Audit phase into system performance requirements.
This phase decomposes the problem into engineering domains and defines requirements for:
- Hardware (HW)
- Firmware (FW)
- Software (SW)
Design activities include defining:
- system architecture
- performance specifications
- subsystem interfaces
- signal processing and algorithms
- wireless communication requirements
- power budgets and form factors
At this stage, the system becomes technically defined and implementable.
Validate
The Validate phase implements the system defined during the Design phase and verifies that it performs according to the original use cases defined during the Audit phase.
Validation includes:
- implementation of HW, FW, and SW
- system integration testing
- performance verification
- reliability testing
- confirmation that the system delivers the intended use-case outcomes
Validation closes the loop between intent, design, and real-world performance.
The Discipline
If use cases defined in the Audit phase are not translated into measurable requirements during the Design phase, and proven during the Validate phase, the result is not a deployable system.
It is only a prototype.
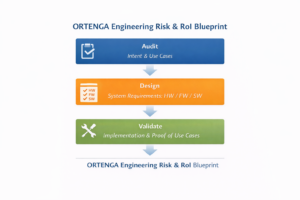
Figure — ORTENGA Engineering Risk & RoI Blueprint
A disciplined Audit → Design → Validate framework aligns engineering execution with business objectives, reducing development risk and protecting return on investment.
Closing Insight
Non-invasive glucose monitoring sits at the intersection of biosensing, electronics, wireless systems, and data analytics.
The companies that succeed will not simply build sensors.
They will build complete, validated systems capable of delivering reliable health data at scale.
Partner with ORTENGA to define, design, and validate glucose detection and measurement platforms that move from prototype to production.